ARTICLE
Tips for turtles: how to improve success when rehabilitating turtles
Sue Carstairs
Ontario Turtle Conservation Centre, Peterborough, ON, Canada
Abstract
Turtles are among the most endangered vertebrates on the planet. While they face many threats, road mortality is one of the main causes for population declines in North America. Due to their life history (extremely long lives, slow maturity and low recruitment into the adult population), adults lost on the roads are impossible to replace in the population. As a result, treatment and rehabilitation of adult turtles is unique in its conservation impact. Turtles are very different than mammals and birds in their response to medical treatment, but their rehabilitation can be very rewarding. This paper outlines some simple approaches to their care that can increase success for rehabilitation of these species.
BIO
Sue Carstairs is a veterinarian with 25 years of wildlife experience. She has been the executive and medical director of the Ontario Turtle Conservation Centre (OTCC; operating name of the Kawartha Turtle Trauma Centre) since 2009. The OTCC runs a teaching and referral hospital admitting approximately 2000 trauma cases annually, a headstarting programme, a long-term headstarting post-release study, as well as an extensive public education programme.
Keywords
Turtle rehabilitation; turtle road mortality; turtle trauma; turtle conservation; Ontario turtles
Citation: Wildlife Rehabilitation Bulletin 2025, 43(1), 12–21, http://dx.doi.org/10.53607/wrb.v43.280
Copyright: Wildlife Rehabilitation Bulletin 2025. © 2025 Sue Carstairs. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), permitting all non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Accepted: 3 February 2025; Published: 1 August 2025
Competing interests and funding: There is no competing interest reported by the author.
There was no specific funding for this paper.
Correspondence: Sue Carstairs, Ontario Turtle Conservation Centre, 2785 Television Road, Douro-Dummer, Ontario, K9L 1E9, E-mail: suecarstairs@sympatico.ca
Introduction
Turtles are among the most endangered group of vertebrates on the planet (Hoffmann et al. 2010). Of the 356 turtle species worldwide, 61% are threatened or have become extinct in modern times (Lovich et al. 2018). Habitat destruction, overharvesting for food and the pet trade, and road mortality, are a few of the major threats that they encounter globally (Gibbons et al. 2000). While they can have exceptionally long lives (Gibbons 1987), very few hatchlings ever make it to adulthood. They are slow to mature but have long reproductive lifespans (Congdon et al. 1994). As a result, turtle populations rely on low adult mortality to maintain population health (Heppell 1998). Adult freshwater turtles often travel many kilometres on land, and as a result invariably encounter roads, with predictable negative results. Both males and females are affected equally (Carstairs et al. 2018), and the losses are considerable. While their life history creates a need for maintaining a stable adult population, this life history also translates into significant positive conservation impacts from rehabilitation of these species (Paterson et al. 2021). As a result, rehabilitation can have a population effect; and can buy time to “fix” the problem with mitigation measures such as eco-passages. Presented here is some basic background information to aid in decision-making on drug administration. Following this, some common cases typically presented to wildlife rehabilitation centres are described. This is of course in no means an exhaustive compilation, but will hopefully help in feeling more comfortable with chelonian cases. Methods described focus on successful outcomes, with modalities that are time expedient, and with low invasiveness, with the motto “why make it complicated?.”
Discussion
Being ectotherms, turtles have a very slow metabolic rate and rely on ambient temperature for all life processes, including healing (Mitchell & Diaz-Figueroa 2004). Healing may progress slowly, but extensive trauma (Fig. 1) often does not lead to mortality as it would for other species due to their protective shell. While of course not all turtles survive, it has proven difficult to rely on prognostic indicators as a way of triaging out hopeless cases. As a result, treatment is attempted for most cases deemed to have a chance at survival, with often remarkable results. While methods described here tend to be fairly simple, there are examples of more complicated and invasive procedures, in the literature. For example, utilising screws implanted in the shell, in order to wire fracture pieces together (Roffey & Miles 2017) and vacuum assisted healing (Lafortune et al. 2005). Simpler methods however, can generally still provide a positive outcome. Vacuum-assisted healing may well be practical and advantageous when dealing with relatively few cases, but when admission numbers are large, or when turtles must be juggled alongside large numbers of other species, as in most rehabilitation settings, this is frequently not feasible. Screw fixation requires increased invasiveness, and time, without any success advantage.

Fig. 1 A typical presentation of vehicle-related injury in a painted turtle. Even though the coelom and organs are exposed, this is a repairable injury. When organs are damaged (e.g., lacerated intestines), it carries a much poorer prognosis, however.
Veterinary expertise is essential in managing these cases, since controlled drugs are frequently used, and sedation or full anaesthesia is normally required, as well as extensive surgical and medical knowledge. In addition, the means to carry out diagnostic procedures is required. However, there is an extensive amount of work that the lay rehabilitator can carry out, in collaboration with, and in preparation for, their veterinarian.
All turtles that have experienced a vehicular injury, will require analgesics. They may not show pain as mammals do, but it is agreed that they can detect and perceive pain (Perry & Navarez 2018). The type of pain receptors present in an organism will determine which analgesic has optimum effect. These receptors differ between reptiles and mammals or birds, and also differ between reptile species. Therefore, extrapolation between species is not possible. As such, drugs such as meperidine, buprenorphine, and butorphanol do not tend to be efficacious in turtles (Serinelli et al. 2022). Likewise, the bioavailability of the particular drug will vary and the dosage frequency for reptiles will not necessarily be the same as with mammals and birds, or even between different species of reptiles.
While the efficacy or dosage of nonsteroidal anti-inflammatories (NSAIDs) such as meloxicam, have not been determined as yet in turtles, it is still used regularly (Serinelli et al. 2022). In fact, the topic of analgesics in reptiles in general, is in its infancy; therefore, it is important to keep up with the literature and change protocols as more is learned. The NSAIDs are not “controlled” and are therefore easier to use and access in a wildlife rehabilitation setting (“controlled” drugs being any type of drug that the federal government has categorised as having a higher-than-average potential for abuse or addiction, and so have restrictions placed on their prescription and use). The deleterious effects of NSAIDs documented in birds and mammals, should be kept in mind when using these medications. Opioids such as hydromorphone, meperidine, methadone, morphine, and tramadol, have been shown to have good analgesic effects in reptiles (Sladky & Mans 2012). However, respiratory depressive effects have only been studied in morphine and tramadol. Both of these offer good analgesia (Serinelli et al. 2022), but tramadol has the advantage in a critical patient, of not causing the respiratory depression that morphine does (Sladky & Mans 2012).
The College of Veterinarians of Ontario stipulates that controlled drugs can only be administered when a licenced veterinarian is actually present on the premises. A veterinarian acting as the veterinarian of record for a wildlife rehabilitation centre, cannot legally prescribe and leave controlled substances for use when the veterinarian is not there in person. Since most rehabilitation centres do not have a veterinarian on full time staff, this limits the feasibility of use of controlled substances in a wildlife rehabilitation setting.
The location of injection site in reptiles has come under much discussion over the years. Reptiles have a “renal portal system” not found in mammals, as well as a “hepatic portal system” that differs from the portal system of mammals. As a result, blood draining the caudal body of reptiles passes through the kidneys or the liver before reaching the central circulation. In the past, it was recommended that all injections be given in the front half of the reptile, with the theory that if they were given in the back end, they would either be excreted before being absorbed, or would increase nephrotoxic or hepatotoxic effects in those drugs with this characteristic. However, the effect will vary with a particular drug’s renal or hepatic excretion rate, and whether they are excreted via glomerular filtration. It has however, been shown that generally this effect is unlikely to be clinically significant (Holz et al. 1997). Nonetheless, it is important to know the excretion route of a particular drug, in order to make an informed decision, as there is much variation between drugs. However, clinical response may not correlate to theoretical response. For example, oxytocin is excreted primarily by the liver; and in theory, injection in the front end should show a superior clinical response, by preventing the hepatic first pass effect. In an unpublished study at Ontario Turtle Conservation Centre (OTCC), however, there was no difference in response seen, when given in the hind end versus the front end. Tramadol effects are actually improved if given in the hind end, as it results in 20% higher amounts of metabolites via this route, which have 200 times the affinity for receptors than the parent drug tramadol (Giorgi et al. 2015). While tramadol was shown to have a slower onset and shorter duration of activity when given via the subcutaneous route versus the oral route (Baker et al. 2011), the study didn’t stipulate where the subcutaneous injection was given. In addition, the oral route carries the disadvantage of logistical challenges with a lot of Ontario’s native turtle species, causing risk of injury to both turtle and handler, as well as potential inaccurate dosing due to regurgitation. At OTCC, generally the subcutaneous route is used, and tramadol is compounded for this at a compounding pharmacy. Generally, the subcutaneous route for drug administration is preferred to the intramuscular route (Sladky & Mans 2012).
Multimodal analgesia is frequently used in mammals and birds, but no studies have been carried out as yet in reptiles (Serinelli et al. 2022). In theory, a combination of centrally and peripherally acting opioids, NSAIDs that act at the tissue level to control inflammatory pain, as well as local anaesthetics that block the transmission of the pain signal, would offer the best advantages. Local anaesthetics such as mepivacaine, lidocaine, and bupivacaine, have not had toxicity studies carried out as yet for reptiles, but are widely used.
A turtle’s carapace is comprised of the ribs and vertebra fused into one casement. There lies a membrane (pleuroperitoneal membrane) below this, and beyond this is the body cavity or coelom. The lungs lie directly under the shell cranially. Fractures through the shell often lead directly into the coelom. Therefore, cleaning of the wound must keep this in mind. The vigorous flushing that is the norm in mammals will often lead to contaminated fluids being tracked into the body cavity, or worse; to drowning. Also, any creams or wound dressings applied will also wind up in the coelom if a shell defect of any size exists. Therefore, initially very conservative flushing of wounds is advised, and if in doubt as to defects leading to the coelom, do not flush. At OTCC, infection of wounds is rarely seen. As a result, antibiotics are used minimally and are reserved initially for wounds clearly older and heavily contaminated. Whether antibiotics are required during the healing process will be determined case by case by daily monitoring. The antibiotic of choice will depend on culture and sensitivity results. In addition, monitoring of the complete blood count (CBC) as well as cell morphology will be an aid for infection detection and monitoring.
A key to successful wound management lies in the daily nursing care and treatment of the wound, plus prevention of contamination with fly eggs and resultant maggots. Fly netting to cover holding containers, and sterile coverings to the wounds, are essential aids to prevent fly invasion. A dry sterile covering such as TelfaTM non-adherent dressing (Cardinal Health, Dublin Ohio) is generally placed initially, and only once it has been confirmed that no tract into the coelom exists, will wound dressings be utilised, as indicated. An adhesive cover, such as HypafixTM (BMS Medical, Des Moines IA) or TegadermTM (3M Health Care, St. Paul, MN) cut to fit over the sterile pad, acts as a good first wound treatment, and prevents fly access.
Emergency care and systemic support for turtles is often extrapolated from that known for mammals and birds, although we are constantly learning more specifically about reptiles. Being ectotherms, it is important to maintain them at their Preferred Optimum Temperature Zone, for ideal absorption of drugs and healing (Mickelson et al. 2016). While mobile turtles are provided with a heat gradient within their enclosure, in the way of a heat and (Ultraviolet B) lamp, newly traumatised individuals tend to be immobile, and not able to move away from a heat source. As a result, care should be taken in providing supplemental heat, to ensure that the turtle is not overheated while it is immobile. In addition, the trauma patient should be kept out of water until mentation is sufficient to prevent drowning, and wounds are properly assessed. Many wounds require being kept dry, especially those with potential tracks into the coelom. If these are present on the carapace, however, often shallow water can be provided once mentation is sufficient, while still allowing dry dressing of wounds. Extensive dry-docking (greater than 7–10 days) is not tolerated well by the species of turtles seen at OTCC, and mentation suffers as a result; therefore, every effort is made to have them exposed to at least shallow water by then.
While dry-docked, it is important to prevent secondary pressure wounds on the plastron, by providing a padded substrate. Thick rubber matting accomplishes this purpose, and all turtles are kept on this while dry. Snapping turtles particularly are prone to ulcerations on their proximal plastron, that can become problematic, thus prevention is the key. “Road rash” on the plastron is common especially in the small species such as the painted turtle, and needs to be addressed on admission, to prevent progression. Generally topical application of a cream addressing gram negative bacteria, applied daily, will accomplish this.
Fluid therapy is used for all trauma patients, and isotonic crystalloid solutions have been shown to be appropriate (Parkinson & Mans 2020). The use of Lactated Ringers Solution was historically not recommended, due to a concern for hyperlactatemia. However, this has not been shown to be the case, and its use is safe (Parkinson & Mans 2020). Because of the large number of admissions at OTCC (often 50–60 a day in the summer season), it is not feasible to maintain patients on intravenous fluid therapy; and instead generally, subcutaneously administered fluid therapy is carried out three times a day. Subcutaneous fluid administration has been shown to be absorbed well (Parkinson & Mans 2020), and is generally the route of choice. Dose and frequency of fluid administration needs to be adjusted based on hydration status, species, and condition.
The main goal during the initial phase of treatment is to support the patient by way of fluids and analgesics, and initial wound stabilisation is done to ease comfort. However, permanent fixation is saved until the turtle’s overall condition allows anaesthesia. For small species such as painted turtles, initial fracture stabilisation can be as simple as placing thin strips of tape across the fracture and at right angles to the fracture site. To allow these to stay in place, a “super glue” containing cyanoacrylate is used to secure the tape to the shell, by placing a few drops at either end of the tape. (e.g., LePage Super Glue Ultra GelTM ControlR, Henkel, Dusseldorf, Germany). Unlike tissue adhesives (which also contain cyanoacrylate but are compatible with tissue), these super glues result in marked acute and chronic tissue reaction (Cascarini & Kumar 2007), and should not be allowed to enter a wound. The gel formulation allows tighter control of application, with less likelihood of it running into the fracture site, and is therefore preferred over the liquid formulation. Permanent fixation of these fractures will require drilling holes at the margin of the shell, under sedation or anaesthesia; utilising a high-speed dental drill, and wiring the fracture sites together with orthopaedic wire, similar to a suture (Fig. 2). These wires are however, only placed at the margins to prevent contamination into the coelom. Marginal wiring is required to ensure good apposition of the fracture sites and secure healing. Tape alone will often not accomplish this, and often will result in mobility of the fracture site and a resultant non-union. While a cable clip and tie method can be utilised in larger turtles such as Blanding’s and snapping turtles, to augment fracture stabilisation in addition to wiring (Fig. 3), in smaller turtles this will provide too much pressure and often will pull the fracture pieces out of alignment, so tape alone is used along with the wiring. Other methods have been described, such as utilising clothing clips and wires to traverse fracture sites (Roffey & Miles 2017). However, in our experience, for the smaller turtles, the simplistic tape method is quicker, and just as effective, with no need to wait for epoxy to dry. Marginal wiring completes the job, and ensures security of fracture sites. However, in facilities without access to dental drills, the clip and wire method may be useful.

Fig. 2 Photo at left depicts initial presentation of painted turtle with carapace fractures. The fracture sites can be initially stabilised with tape secured with glue at each end, and later fully fixated utilising orthopaedic wire securing the fracture pieces (arrows). A dental drill is used, to drill small holes through which to feed the orthopaedic wire. Only the edges of the carapace are utilised for this method. While fractures affecting the spinal cord do carry a poorer prognosis, varying degrees of spinal regeneration is possible, justifying repair attempts for the majority of these cases, since many do go on to full function.

Fig. 3 Cable clip and cable tie method to augment fracture stabilisation. Cable clips are attached to the shell using quick-drying epoxy, and two cable ties are utilized to force the fracture pieces together. The shell margins are still wired utilising the high-speed dental drill and orthopaedic wire as seen in Fig. 1.
Because these wounds are often contaminated, we do not recommend covering the wound with a permanent “patch” as visualisation of the wound on a daily basis is needed, in order to quickly identify infection or maggot infestation. The only time a “patch” would be of benefit would be in a case with extensive defects into the coelom, that are not resolved within 7–10 days, and that do not allow access to even shallow water.
The manipulation of the fracture segments generally requires administration of at least short acting anaesthesia. Choice of product to use will be determined by veterinary preference. Alfaxalone (AlfaxanR Zoetis, Parsippany, NJ) is a good alternative for short procedures or for induction for longer procedures. It is a neuroactive steroid and general anaesthetic, that is currently not controlled in Canada, but is considered a Class IV controlled substance in the US. The intravenous route gives the quickest and most reliable onset of anaesthesia, but it can be given subcutaneously as well. However, it does not have the reversal benefit of some other anaesthetics. The dose of Alfaxalone to use for sedation or short acting anaesthesia, will vary greatly between species. Therefore, it is important to be familiar with the species differences prior to use, to prevent under or over-dosing.
With carapace fractures, there is often a degree of spinal trauma. It is important not to utilise the same criteria for assessing these as with mammals or birds, however, since freshwater turtles have been found to be able to reconnect their completely transected spinal cords (Rehermann et al. 2009). Research has shown that the end result is variable, and the cases at OTCC corroborate this. While some may not regain sufficient function for release, enough do, to warrant supportive treatment. Ongoing research should shed more light on this topic.
Often pieces of shell will be missing, and this results in an open hole directly into the coelom (Fig. 4). The lack of a diaphragm allows the turtle to still be able to breath in this situation, unlike in mammals. It is very important that these wounds be dressed daily with sterile dressing to prevent secondary problems. The wounds will invariably heal very well, even if quite large, secondary complications can lead to life threatening problems. Adhesive dressings are important, to cover a sterile dressing and allow no access for flies. Holes at the bridge are particularly common in painted turtles, and holes scraped into the dorsum of the carapace are common in the larger snapping turtles. Patience is important, in treating these wounds.

Fig. 4 Defects in the shell (left) will heal well with good nursing care. The photo on the right shows the same defect once fully sealed. The wound is kept covered during this time and the turtle is dry-docked. If the wound is still open after 10 days or so, generally a waterproof temporary patch is placed so that the turtle can be exposed to at least shallow water.
Multiple fractures leading to many small, indented fragments, should be repositioned as best as possible, while attempting to maintain the integrity of the pleuroperitoneal membrane where possible. It is sometimes preferable to have an imperfect wound site that is with the pieces indented, rather than causing more damage by attempting to perfectly position them.
Facial trauma is most common in the snapping turtles (Figs. 5–7). Their inability to retract completely into their shell is the likely reason. Their size and resilience translate into higher survival with these types of injuries than with smaller turtles. As a result, they often are admitted with fractures to the maxilla and/or mandible, and with severe head and facial trauma. The wounds themselves can generally be treated and will heal, despite horrendous first appearances. Fractures of the maxilla or mandible with pieces large enough to do so, are best apposed with wire (Fig. 8). A full general anaesthesia is of course necessary to accomplish this. Trauma resulting in many fractures, or missing pieces are still amenable to treatment; however, the method will be on a case-by-case basis. Often, it is a matter of realigning the pieces and securing with small strips of tape and tissue adhesive. Mentation will vary with individuals, but snapping turtles often have normal mentation even with severe head trauma, due to their very tough skulls.

Fig. 5 Facial trauma in a snapping turtle, repaired with tape (middle), since it was not amenable to wires or suture. Far right shows the same turtle shortly before release.
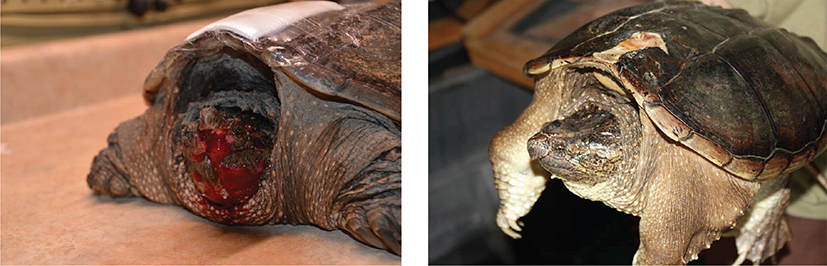
Fig. 6 It can sometimes be difficult to see the extent of injury of facial traumas, on first admission. The photo at left suggests irreparable damage, and yet the same turtle at right shows that both eyes were viable, and the mouth was aligned and fully functional. The turtle was released after healing.
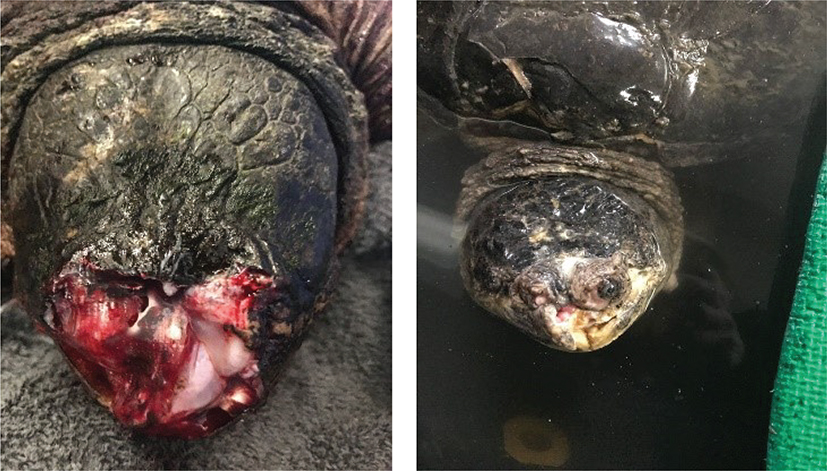
Fig. 7 On admission, it was difficult to determine whether this injury was compatible with life (left). After 6 months of healing via second intention (right), although disfigured, the turtle had full vision in the left eye, and could prehend food well. When the tissue toughened up, he was released.
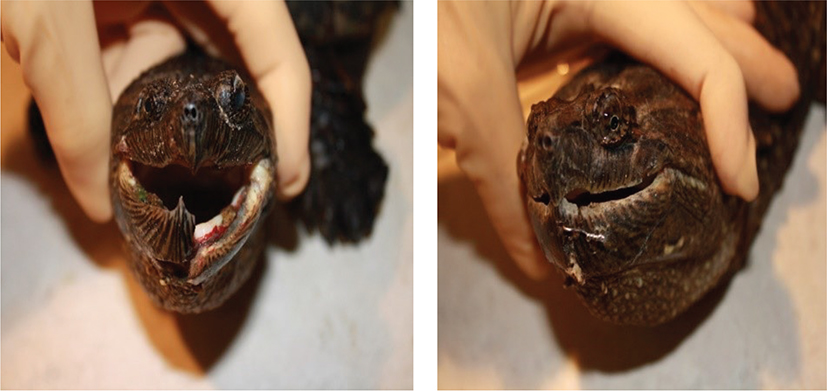
Fig. 8 A mandibular fracture that lends itself well to wiring. The photo in the right shows the same turtles after surgical repair using a dental drill to create holes to feed orthopaedic wire through. The anaesthesia for this must be quite deep.
It is not unusual for trauma cases to be anorectic initially. Body weight and condition should be monitored closely, in order to assess when assisted feeding is necessary. Nutritional supplementation can be added as needed, and once the turtle’s condition allows. While, historically, we utilised indwelling esophagostomy feeding tubes for this, we often find now that delivery via mouth with a rigid feeding tube, is preferable. Since the turtles we admit generally will only eat in water, this allows them to remain in water and so can still be offered normal food as well. With practice, the species we admit, can be tube fed awake quickly, safely, and non-invasively. For snapping turtles, however, occasionally the turtle will be too fractious for tube feeding, leading to danger for turtle or human. The method of assisted feeding will depend on the species seen, the availability of veterinary expertise, and the number of cases.
Deep scraping wounds on the dorsum of snapping turtles (Figs. 9 and 10) heal well when treated as open wounds and dressed daily. Dressing will depend on the wound, and initially a dry sterile dressing is preferred.
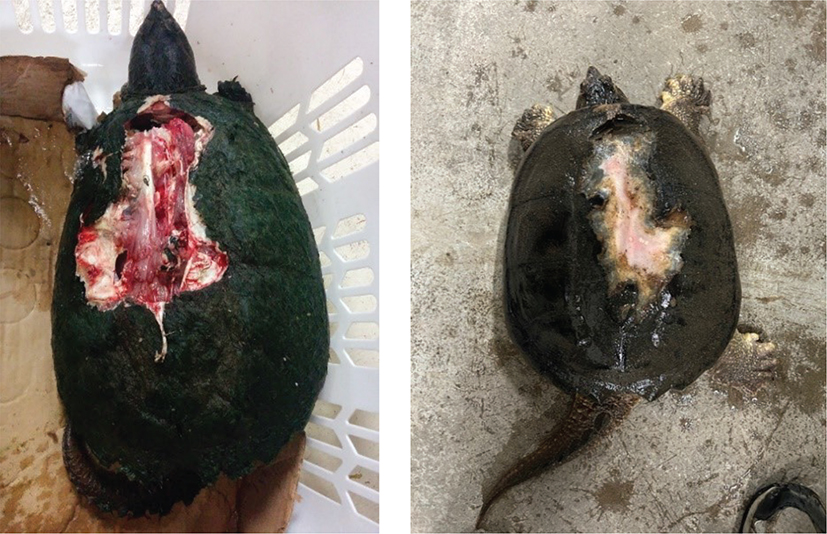
Fig. 9 Typical dorsal deep scraping wounds found in snapping turtles, straddled by a car (left), carry a good prognosis even when they extend into the coelom. With excellent nursing care and wound management, the wounds will heal well (right). Turtles can generally be exposed to at least shallow water in the early stages, as long as no defects exist ventrally. Wounds are kept covered and are dressed daily.
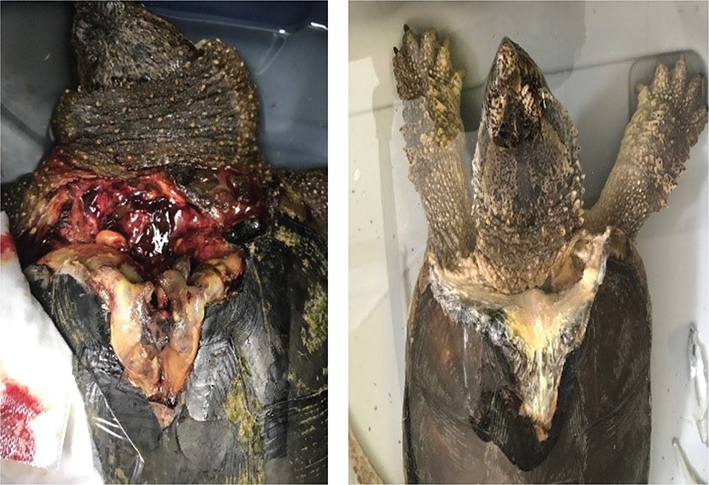
Fig. 10 Left: Initial presentation of a severe traumatic wound of the cranial carapace and soft tissue of the region. Notice the marked edema of the neck. While the turtle found it difficult to raise his head during this phase and needed to be kept in very shallow water as a result, he regained full function of the related muscles. Above right shows the wound after 6 months. The turtle was fully ambulatory, had excellent mentation, and was released.
Fishhook ingestion is a common problem in turtles of all sizes, but particularly in snapping turtles. We often find it as an incidental finding when x-raying for trauma. Fish hook removal does not follow any one rule, and each case must be treated differently. Quite often, hooks that are beyond the stomach will pass independently. Often, however, they lodge at the base of a snapping turtle’s throat, and will require removal. It is still important to treat the initial trauma first, however, even if they came in with this as a secondary problem. Fish hook removal is often more difficult than initial appearances. The voluminous property of their esophagus and high frequency of multiple barbs, makes removal very difficult even when they are accessible. Full anaesthesia and maintenance on inhaled anaesthetic agent, is necessary, to allow time for the surgery. Some hooks can be retrieved via the mouth and often the hook will need to be cut to allow retraction. Other hooks that have settled further down, can be palpated externally. While removal directly through the esophagus to the outside can be possible in these cases, it must be done with great care to avoid the large vessels in this area.
Other presentations
The above-stated descriptions are of course not exhaustive, and there are many different presentations for trauma cases. In addition, a full physical examination should be performed, to look for signs of infectious illness. Appropriate diagnostics should always be carried out, to track these additional threats. In Ontario, there is a high prevalence of asymptomatic Ranavirus infection (Carstairs et al. 2020) and a regular but so far, low prevalence of symptomatic patients. In addition, herpesvirus is seen on a regular basis. Other presentations include aural abscesses particularly in painted turtles (Carstairs et al. 2022).
A note about anaesthesia
Often the most difficult part of surgical treatment of turtles will be the anaesthesia. It is not advisable to tackle this without extensive experience since it can be extremely challenging and frustrating. Since they breath-hold for extended periods, respiratory rate cannot be utilised as a monitoring means. Also, this precludes the use of masks to induce with an inhaled agent. Any inhaled agent will require intermittent partial pressure ventilation (IPPV) constantly, and knowledge of their unique anatomy and physiology to accomplish this successfully (e.g., rigid tracheal rings, necessitating an uncuffed tube to prevent tracheal trauma, short distance to tracheal bifurcation requiring shorter tube used than mammals, etc.). In addition, while heart rates can be monitored, they cannot be relied on to illustrate depth of anaesthesia, since they can have an active heartbeat for many hours after death.
A note about euthanasia
While luckily not necessary very often, there is a time and place for humane euthanasia. A veterinarian will be required for this, since controlled substances need to be utilised. Humane death is the goal and can be accomplished via a two-step method utilising full anaesthesia followed by intravenous sodium pentobarbital (Nevarez 2019). Sodium pentobarbital should not be given intracoelomically unless the patient is fully anaesthetised, as this causes pain (Nevarez 2019). “T-61,” which is often used in a wildlife rehabilitation setting, cannot be humanely used in reptiles. While in dogs and rabbits, it has been shown to accomplish unconsciousness before paralysis of muscles for breathing, this is not the case in reptiles, therefore potentially leading to a very inhumane death. Likewise, since brain activity can continue for a significant time after decapitation, this is also not a humane method. Freezing is also not considered a humane method of euthanasia, combined with the fact that some species can withstand freezing and thawing. While “MS 222” has been recommended by some, it has not been proven to be a humane euthanasia method (Nevarez 2019).
References
| Baker B.B., Sladky K.K. & Johnson S.M. 2011. Evaluation of the analgesic effects of oral and subcutaneous tramadol administration in red-eared slider turtles. Journal of the American Veterinary Medical Association 238, 220–227, doi: 10.2460/javma.238.2.220. |
| Carstairs S., Dupuis-Desormeaux M. & Davy C.M. 2018. Revisiting the hypothesis of sex-biased turtle road mortality. Canadian Field-Naturalist 132(3), 289–295, doi: 10.22621/cfn.v132i3.1908. |
| Carstairs S.J., Dutton C.J. & Dupuis-Desormeaux M. 2022. Aural abscesses in wild midland painted turtles (Chrysemys picta) admitted to a wildlife hospital in Ontario, Canada 2011–2020. Journal of Wildlife Diseases 58(2), 389–393, doi: 10.7589/JWD-D-21-00067. |
| Carstairs S.J., Kyle C.J. & Torres Vilaca S. 2020. High prevalence of subclinical frog virus 3 infection in freshwater turtles of Ontario, Canada. Virology 543, 76–83, doi: 10.1016/j.virol.2020.01.016. |
| Cascarini L. & Kumar A. 2007. Emergency casebook case of the month: honey I glued the kids: tissue adhesives are not the same as “superglue.” Journal of Emergency Medicine 24, 228–231, doi: 10.1136/emj.2006.036392. |
| Congdon J.D., Dunham A.E. & van Loben Sels R.C. 1994. Demographics of common snapping turtles (Chelydra serpentina): implications for conservation and management of long-lived organisms. American Zoologist 34, 397–408, doi: 10.1093/icb/34.3.397. |
| Gibbons J.W. 1987. Why do turtles live so long? BioScience 37, 262–269, doi: 10.2307/1310589. |
| Gibbons J.W., Scott D.E., Ryan T.J., Buhlmann K.A., Tuberville T.D., Metts B.S., Greene J.L, Mills T., Leiden Y., Poppy S. & Winne C.T. 2000. The global decline of reptiles, déjà vu amphibians. BioScience 50, 653–666, doi: 10.1641/0006-3568(2000)050[0653:TGDORD]2.0.CO;2. |
| Giorgi M., Salvadori M., De Vito V., Owen H., Demontis M.P. & Varoni M.V. 2015. Pharmacokinetic/pharmacodynamic assessments of 10 mg/kg tramadol intramuscular injection in yellow-bellied slider turtles (Trachemys scripta scripta). Journal of Pharmacology and Therapeutics 38(5), 488–496, doi:10.1111/jvp.12206. |
| Heppell S.S. 1998. Application of life-history theory and population model analysis to turtle conservation. Copeia 1998, 367–375, doi: 10.2307/1447430. |
| Hoffmann M., Hilton-Taylor C., Angulo A., Bohm M., Brooks T.M., Butchart S.H.M., Carpenter K.E., Chanson J., Collen B. & Stuart S.N. 2010. The impact of conservation on the status of the world’s vertebrates. Science 330, 1503–1509, doi: 10.1126/science.1194442. |
| Holz P., Barker I.K, Burger J.P., Crawshaw G.J. & Conlon P.D. 1997. The effect of the renal portal system on pharmacokinetic parameters in the red-eared slider (Trachemys scripta elegans). Journal of Zoo and Wildlife Medicine 28(4), 386–393. |
| Lafortune M., Wellehan J.F.X., Heard E., Rooney-Del Pino D.J. & Fiorello C.V. 2005. Vacuum-assisted closure (Turtle VAC) in the management of traumatic shell defects in chelonians. Journal of Herpetological Medicine and Surgery 15(4), 4–8, doi: 10.5818/1529-9651.15.4.4. |
| Lovich J.E, Ennen J.R., Agha M. & Whitfield J. 2018. Where have all the turtles gone and why does it matter? BioScience 68, 771–781, doi: 10.1093/biosci/biy095. |
| Mickelson M., Mans A.C. & Colopy S.A. 2016. Principles of wound management and wound healing in the exotic pets. Veterinary Clinics of North America Exotic Animal Practice 19(1), 33–53, doi: 10.1016/j.cvex.2015.08.002. |
| Mitchell M.A. & Diaz-Figueroa O. 2004. Wound management in reptiles. Veterinary Clinics of North America Exotic Animal Practice 7(1), 123–140, doi: 10.1016/j.cvex2003.08.006. |
| Nevarez J.G. 2019. Euthanasia. In S.J. Divers & S.J. Stahl (eds.): Mader’s reptile medicine and surgery. Pp. 437–440. 3rd edition. St. Louis, MO: Elsevier. |
| Parkinson L.A. & Mans C. 2020. Evaluation of subcutaneously administered electrolyte solutions in experimentally dehydrated inland bearded dragons (Pogona vitticeps). American Journal of Veterinary Research 81(5), 437–441, doi:10.2460/ajvr.81.5.437. |
| Paterson J.E., Carstairs S.J. & Davy C.M. 2021. Population-level effects of wildlife rehabilitation and release vary with life-history strategy. Journal of Conservation of Nature 61, 128083, doi: 10.1016/j.jnc.2021.128083. |
| Perry S.M. & Nevarez J.G. 2018. Pain and its control in reptiles. Veterinary Clinics: Exotic Animal Practice 21(1), 1–16, doi: 10.1016/j.cvex.2017.08.001. |
| Rehermann M.I., Marichal N., Russo R.E. & Trujillo-Cenóz O. 2009. Neural reconnection in the transected spinal cord of the freshwater turtle Trachemys dorbignyi. Journal of Comparative Neurology 515, 197–214, doi: 10.1002/cne.22061. |
| Roffey J. & Miles S. 2017. Turtle shell repair. In B. Doneley et al. (eds.): Reptile medicine and surgery in clinical practice. Pp. 397–408. Hoboken, NJ: John Wiley and Sons. |
| Serinelli I., Soloperto S. & Lai O.R. 2022. Pain and pain management in sea turtle and herpetological medicine: state of the art. Animals 12, 697, doi:10.3390/ani12060697. |
| Sladky K.K. & Mans C. 2012. Clinical anesthesia in reptiles. Journal of Exotic Pet Medicine 21(1), 17–31, doi: 10.1053/j.jepm.2011.11.013. |