ARTICLE
Surfactant efficacy in removal of petrochemicals from feathers: results using an improved method for objective testing
Andrea Martin, Jada Pagan, Noah Smeriglio & Tyler Apple
Chemistry Department, Widener University, Chester, PA, USA
Abstract
A set of detergents were tested in a blind study to determine their efficacy in removing a hydrocarbon blend from feathers. In the course of the work, significant modifications were made to the published procedure to improve precision and reduce cost. The most effective detergents were found to be Palmolive Ultra®, Dawn Ultra Pure Essentials® and its UK equivalent, Fairy Liquid, and Bright GreenTM.
BIO
Andrea Martin is Emeritus Professor of Chemistry at Widener University, where she taught a range of chemistry courses and carried out an active undergraduate research program. She is a member of the American Chemical Society and the Alpha Chi Sigma Professional Chemistry Fraternity. She is an avid birder in her spare time. Email: aemartin1@widener.edu
Jada Pagan is a junior chemistry major at Widener University with an interest in a health-care profession after graduation. She presented her portion of this work at the NRWA symposium in Wilmington, DE, in March 2023.
Noah Smeriglio graduated from Widener University with a dual degree in chemistry and chemical engineering in May 2022. He is currently a graduate student in chemistry at George Washington University.
Tyler Apple graduated from Widener University with a dual degree in chemistry and chemical engineering in May 2022. He is currently a chemist with Eurofins.
Keywords
Surfactant; petrochemical; oiled wildlife
Abbreviations
GC: gas chromatography
RSD: relative standard deviation
TSBRR: Tri-State Bird Rescue & Research
NWRA: National Wildlife Rehabilitators Association
Citation: Wildlife Rehabilitation Bulletin 2025, 43(1), 38–47, http://dx.doi.org/10.53607/wrb.v43.277
Copyright: Wildlife Rehabilitation Bulletin 2025. © 2025 Andrea Martin et al. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), permitting all non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Accepted: 26 January 2025; Published: 1 August 2025
Competing interests and funding: The authors report no conflict of interest.
The authors thank the Oiled Wildlife Care Network for substantial funding for this project. We also thank Tri-State Bird Rescue & Research, Widener University, and Hamilton Syringe for the financial support.
Correspondence: Andrea Martin, Professor Emeritus, Chemistry Department, Widener University, 1 University Place, Chester, PA 19013. E-mail: aemartin1@widener.edu
To access the supplementary material, please visit the article landing page
Introduction
Petroleum products form a major part of the world’s economy, mainly as a source of energy. Annual worldwide usage in 2021 was nearly 100 million barrels per day, not only for fuel but also for plastics, coatings and consumer products (U.S. Energy Information Agency 2023). In the course of production and transportation, spills are nearly inevitable, resulting in significant environmental impact. It is estimated that over one million birds ultimately died as a result of the Deepwater Horizon spill in the Gulf of Mexico in 2010 (Audubon 2014). In addition to birds, sea turtles, marine and terrestrial mammals, invertebrates and plants are at risk from oil spills (Center for Biological Diversity, undated). The effects of exposure to petroleum products on birds have been well documented (King et al. 2020), including disrupting feather structure, affecting buoyancy, flight, thermoregulation and systemic toxicity from ingestion and inhalation. Other animals are similarly affected.
Since the 1970s, a number of organizations have sprung up worldwide to respond to the aftermath of oil spills, including Tri-State Bird Rescue & Research in Delaware and the Oiled Wildlife Care Network in California. These organizations treat affected wildlife with the goal of rehabilitating them and returning them to the wild. To support these efforts, TSBRR partnered with DuPont to develop both qualitative (subjective) and quantitative (objective) methods for testing potential cleaning agents (Bryndza et al. 1991). Periodic retesting of detergent efficacy has been conducted (Bryndza et al. 1995; Miller et al. 2003; Miller et al. 2006; Miller & Ricko 2022). These studies have consistently identified formulations of Dawn® and Palmolive® dish detergents as the most effective based on the quantity of residual oil remaining on washed feathers. As noted previously (Miller & Ricko 2022), effective detergents must remove petroleum products at physiological temperatures and rinse cleanly without toxic effects to the animal. Moreover, they must be readily available in a variety of locations where wildlife may be contaminated.
The purpose of this study was to replicate the original procedure (Bryndza et al. 1991) with a set of unknown detergents provided by TSBRR. However, significant difficulties were encountered when attempting to reproduce the published procedure. Consequently, several modifications were made, leading to the development of an improved testing method, which was then used to evaluate the detergents. The revised process for detergent testing improves precision and reduces the quantity of toxic solvents employed at a significantly lower cost compared to the original method. In a blind study using the new method, the most efficacious detergents were found to be Palmolive Ultra®, Dawn® Ultra Pure Essentials and the UK equivalent of Dawn, Fairy Liquid, and Bright GreenTM.
Materials and methods
Eleven cleaning products were provided by TSBRR in 2020, labelled A through K. The identity of the detergents was revealed after testing was completed. By the time testing was completed in 2022, it is possible, but highly unlikely, that degradation of the detergents had occurred. Product information for these detergents is given in Table 1. Hydrocarbons used to prepare synthetic analogues of light crude oil, solvents (dichloromethane and acetone) and anhydrous potassium carbonate were obtained from Sigma-Aldrich (St. Louis, MO) or Fisher Scientific (Hampton, NH) and were used without purification. Snow goose (Anser caerulescens) contour feathers were provided under federal licence from TSBRR from carcasses that had been seized from illegal hunting or from birds that had been humanely euthanized whilst in rehabilitation.
| TSBRR ID | Product | Manufacturer | Location |
| A | Dawn® Ultra Pure Essentials | Procter & Gamble | Cincinnati, OH 45202, USA |
| B | Palmolive Pure & Clear® | Colgate-Palmolive Company | New York, NY 10022, USA |
| C | Bright GreenTM * | Safeway | Phoenix, AZ 85038 |
| D | Dawn® Platinum Powerwash | Procter & Gamble | Cincinnati, OH 45202, USA |
| E | Homesolv® by CitraSolv | HomesolveTM, LLC | Danbury, CT 06813, USA |
| F | Charlie’s Soap® | Charlie’s Soap®, Div. Sutherland Products, Inc. | Stoneville, NC 27048, USA |
| G | Fairy Liquid | Procter & Gamble | West Thurrock, England, UK |
| H | Dr. Bronner’s 18-in-1 Hemp Baby Unscented Pure Castile Soap | Dr. Bronner’s | Vista, CA 92081, USA |
| I | Palmolive Ultra® | Colgate-Palmolive Company | New York, NY 10022, USA |
| J | Seventh GenerationTM | Seventh GenerationTM | Burlington, VT 05401, USA |
| K | Ultra Joy® | Procter & Gamble | Cincinnati, OH 45202, USA |
| *Note: This product is no longer available under this tradename but has been rolled into the Albertsons Companies “Open Nature” family of cleaning products. It is unknown how these detergents will compare with the Bright Green formulation. | |||
The published method (Bryndza et al. 1991) involved oiling feathers with a well-defined mixture of equal masses of 13 hydrocarbons (see Table 2), followed by shaking with detergent solutions. After the wash, the feathers were shaken successively with two portions of water as a rinse. Oil remaining on the feathers after washing and rinsing was then extracted with a solvent containing an internal standard for analysis by GC. The mass of each hydrocarbon remaining on the feather sample was determined by the ratio of the peak area of that component compared to that of the internal standard, using instrument response factors determined by analysis of a known mixture. These values were then summed to calculate the total mass of oil remaining after treatment. Controls were obtained by “washing” oiled feathers with warm tap water three times without detergent.
In the original protocol (Bryndza et al. 1991), the hydrocarbon mixture was applied to the feathers as a 50/50 (v/v) solution in dichloromethane. The feathers were shaken with the oil mixture for two minutes, allowed to soak in the mixture for one hour, and then the excess liquid was removed by decantation. Before washing the oiled feathers with the unknown detergents, the solvent was removed by vacuum drying at ambient temperature. The authors indicated that 30 minutes of vacuum resulted in a constant mass of oiled feathers with minimal loss of most of the individual hydrocarbons. Dried feathers were aged overnight (presumably at least 16 hours) in sealed bags.
After ageing, the oiled feathers were weighed into 4-ounce glass jars in 2-g portions and shaken with 10 mL of detergent solutions of various concentrations in tap water at 40°C. The detergent was decanted from the feathers, which were then rinsed by shaking with 10 mL of clean 40°C tap water for 30 seconds. The rinse water was discarded, and the rinse step was repeated. Next, the feathers were filtered from the solution and extracted sequentially with 10 mL acetone (to remove water) and 50 mL dichloromethane containing 1-octadecene as an internal standard for GC. The combined extracts were treated with an inert drying agent, anhydrous magnesium sulphate, to remove any remaining water and then concentrated under vacuum prior to analysis by GC (this last concentration step was eliminated in later studies). The total mass of hydrocarbons remaining on the washed and rinsed feathers was determined in triplicate trials. The authors reported that recovery was within 10% for most components of the mixture, but they noted that the most volatile component, 2-methylheptane (iso-octane), was lost in greater amounts and, therefore, was not reliably measured.
In the course of the current work, significant modifications of the published method were needed to obtain reproducible results. The revised procedure is presented here, and an explanation of the changes is provided in the discussion.
A modified synthetic crude oil blend containing approximately equal amounts of nine substances was prepared as shown in Table 3. Naphthalene and eicosane are solids at room temperature but dissolved readily in the mixture. Due to limited availability, a few of the hydrocarbons were used in a lesser amount, which proved to have no effect on the results. Analysis of the residual oils indicated that substances were recovered in direct proportion to the mass in the synthetic oil blend.
An extraction solution was prepared in a 500 mL volumetric flask by mixing 1.90 g 1-octadecene, an internal standard for GC, in sufficient dichloromethane (methylene chloride) to fill to the mark. To determine the optimum separation conditions and GC response factor for each component of the synthetic blend, a standard sample was prepared with 75.0 µL of the synthetic blend dissolved in enough extraction solution to make a total volume of 2.00 mL.
Oiling feathers
A sample of snow goose feathers (approximately 16 g) was oiled with 500 mL of the hydrocarbon blend described earlier. For convenience, the feathers and oil were divided equally between two 500 mL wide-mouth glass jars. At intervals, the jars were gently rotated to distribute the oil. The feathers were allowed to stand in the oil for a period of at least two hours but not longer than overnight (18 hours). Unless specified otherwise below, the feathers were aged in oil overnight. The oil was then decanted from the feathers and collected. Analysis of the oil showed the composition was unchanged after soaking the feathers, so it was collected and saved for reuse in later experiments.
The oiled feathers were blotted to remove excess oil and then were stored in plastic zip-lock bags to age for 24–72 hours. At this point, 2.00 ± 0.08 g feather samples were weighed, the mass recorded and the feathers placed into individual 2-oz wide-mouth glass jars for washing. The jars were selected at random for triplicate trials for the control and for each detergent.
Uniformity of oiling
To qualitatively test uniformity of oiling using the direct oiling method, a small quantity of fluorescein, a fluorescent dye, was added to a portion of the hydrocarbon blend. Feathers were gently mixed with the dyed blend over a period of a few minutes. The feathers were removed from the blend and observed under visible and UV light. Neither bare spots nor clumps of oil were observed on the feathers, suggesting the oil coating was indeed uniform. Further proof that oiling was uniform is found in the small RSDs of residual oil mass on the cleaned feathers (see later).
Washing and rinsing feathers
The 1% (v/v, with tap water) detergent solutions were prepared and stored in plastic bottles. The solutions were equilibrated to 40°C in a water bath prior to use to approximate the temperature at which oiled birds would normally be washed (Berkner et al. 1977). Throughout this paper, “warm” refers to water and detergent solutions maintained at 40°C. To wash the feathers, 20 mL of the warm detergent solution was placed into the jar with the feathers, and the jar was capped. The mixture was shaken vigorously for 30 seconds and then decanted. To rinse, 20 mL of warm tap water was added to the jar, shaken vigorously for 30 seconds and then decanted. The rinse step was repeated once. The wash and rinse solutions were discarded. For the control, the oiled feathers were shaken with three 20 mL portions of warm tap water (no detergent) and decanted after each treatment with water.
Extraction of residual oil
Ten mL of acetone was added to each jar containing the washed and rinsed feathers. The jar was capped and shaken vigorously for 60 seconds. The acetone was removed by decanting into a filter flask. Next, 50.0 mL of the dichloromethane solution containing 0.2 mg/mL 1-octadecene was added to the feathers and shaken for 60 seconds. The contents of the jar were poured into a glass fritted funnel, and the dichloromethane was suctioned into the acetone solution. Finally, 5 mL of pure dichloromethane was used to rinse out the jar; this was poured over the feathers in the filter, to combine it with the other extracts.
To remove residual water before GC analysis, the combined extract was treated with sufficient anhydrous potassium carbonate until it was no longer hazy (approximately 2 g K2CO3). A 2.0 mL sample of the treated extract was then placed into a GC vial and sealed for analysis. GC analysis was completed within 24 hours of preparing the vial. Instrument conditions are provided in the Supplemental Information.
Three sets of feathers were tested using the modified method described herein, with the results shown in Table 4. These oiled feathers were selected randomly from the complete set of treated feathers prepared for surfactant testing. In most of the previous work (Bryndza et al. 1991; Bryndza et al. 1995; Miller et al. 2003; Miller et al. 2006), RSD of the components averaged less than 10% of the mean value, though the most recent work (Miller & Ricko 2022) was a little higher, at 12.5%. By changing the composition of the blend and eliminating the solvent and vacuum steps, the RSD was reduced to an average of 6%. This low value is conclusive proof that feathers can be uniformly oiled by the direct oiling method without use of a solvent.
The control was repeated on a fresh set of feathers using the oil recovered from the first study (Table 5). Whilst the total oil on the feathers was slightly lower (1300 mg vs. 1500 mg), the uniformity of oiling was confirmed. The lower amount of oil is likely due to a shorter soaking period for the feathers in the oil (about 2 hours vs. about 18 hours) due to time constraints. There appears to be no need to soak the feathers longer than 2 hours, but at the same time, allowing them to soak overnight is not harmful. Importantly, this study confirms that the synthetic crude oil blend can be re-used in multiple trials.
Results
The amount of oil remaining on the washed and rinsed feathers is shown in Fig. 1, which combines the results of testing over two trials on different days. A second trial (shown in orange) was necessary to test all the detergents provided, due to insufficient mass of oiled feathers in the first trial. In the first trial, Dawn Platinum Powerwash was only tested in duplicate, and Seventh Generation was not tested at all. In the second trial, those detergents were tested in triplicate, along with two other detergents chosen at random, with a single control. This provided the opportunity to determine the reproducibility of the method. The error bars indicate one standard deviation. There is good agreement between samples of the same detergent run on different days.
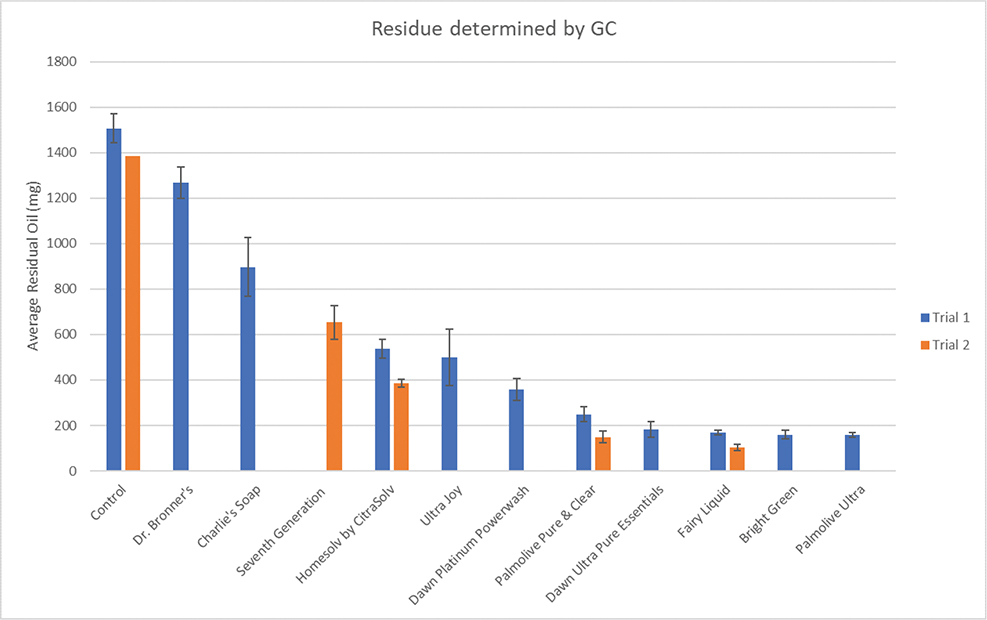
Fig. 1 Residual oil remaining after washing and rinsing.
Control feathers averaged about 1.5 g of oil per 2.0 g of oiled feathers. This is consistent with the original work (Bryndza et al. 1991). There is a clear differentiation between the most and least effective detergents. Palmolive Ultra®, Fairy Liquid/Dawn Ultra Pure Essentials® and Bright GreenTM all removed more than 85% of the oil, whilst, unfortunately, many of the detergents promoted as environmentally friendly showed poor efficacy.
Discussion
The original testing procedure involved treating snow goose contour feathers with a solution of a well-defined hydrocarbon mixture intended to simulate light crude oil, which penetrates feathers and is difficult to remove by washing (Bryndza et al. 1991). The composition of the original mixture is shown in Table 2. It contains the major components that are normally found in crude oil: paraffins, aromatics and naphthenics. By mass, the composition of this blend is 46.2% paraffinic, 38.5% aromatic and 15.4% naphthenic. Paraffins contain only carbon-carbon single bonds, whilst aromatics and naphthenics contain unsaturated benzene rings, the difference being that naphthenics are based on two fused aromatic rings. Our analysis of two authentic light crude oil samples, kindly provided by Monroe Energy, indicated that the synthetic blend contains a significantly higher percentage of aromatics and naphthenics. Both in the prior work and in this study, all three types of hydrocarbons were found in the extracts in proportion to their original concentrations, indicating that whilst the blend does not accurately represent light crude, it is nevertheless useful for testing detergents, which may be used to clean wildlife fouled with other types of oil as well.
Two significant changes to the published procedure were required to obtain reproducible results. The first issue was loss of the costly hydrocarbon blend under vacuum. Thirty minutes of vacuum was insufficient to result in constant mass, but even in that time, a substantial portion of the hydrocarbon blend was lost.
To determine the loss for the individual components, a sample of the published hydrocarbon blend was subjected to vacuum at ambient temperature, and samples were taken at 15 minute intervals for analysis. Figure 2 shows the percentage of each component remaining as a function of vacuum time. The apparent increase in concentration at 15 and 45 minutes is an artefact due to experimental error. Whilst most of the components were constant within +/-10% error, three of the components were lost in excess of 10% within 30 minutes, with 2-methylheptane and ethylcyclohexane lost in greater than about 30%. Further application of vacuum in an attempt to reach constant mass affected several other components as well.
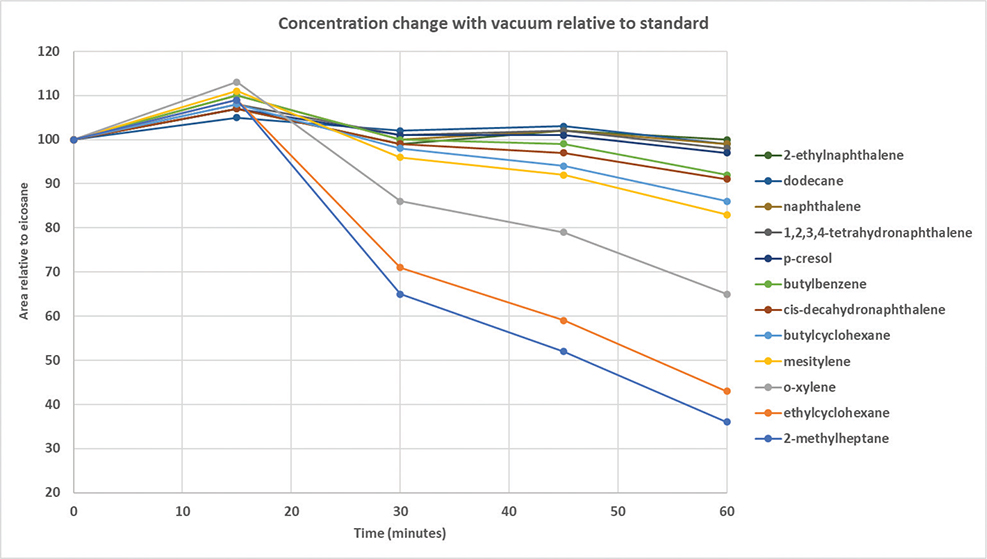
Fig. 2 Loss of hydrocarbons under vacuum as a function of time.
Our best effort to replicate the published work limited vacuum to 30 minutes without any attempt to confirm constant mass, whilst, at the same time, omitting the most volatile components: 2-methylheptane, ethylcyclohexane, o-xylene and n-butylcyclohexane. Even so, after vacuum, 2.0 g of oiled feathers contained less than 50 mg of oil, only about 3% of the 1500 mg expected, leading to high RSD (25%) and low confidence in the results. Due to the unexpectedly low mass of treated feathers, we were unable to test all the detergents provided using this method. In other words, it would have required about 30 times more feathers to be oiled than were available in order to test all the detergents at the expected level of confidence.
It was apparent that the use of a vacuum step caused unacceptable loss of the costly oil blend. To address this problem, feathers were soaked directly in the published hydrocarbon blend (minus the n-butylcyclohexane, which was unavailable from the supplier). Despite eliminating the vacuum step, a complete set of detergent testing with these feathers again resulted in an RSD of about 25%. Analysis of the recovery of each component indicated that greater than 30% of the masses of 2-methylheptane, ethylcyclohexane and p-cresol were lost even without application of vacuum. The most volatile components were generally the most expensive of the components. Hence, the second major modification was to change the blend to remove the most volatile components whilst maintaining a similar proportion of paraffinic hydrocarbons (about 45%). The composition of the revised blend is shown in Table 6. As noted above, the exact ratio of the blend does not appear to be significant, since all of the hydrocarbons tested were recovered in proportion to their amounts, indicating similar solubility in a given detergent. An additional benefit from this modification is that the composition of the decanted hydrocarbon blend remains unchanged after soaking feathers (see below), which allows it to be reused in subsequent trials. Table 7 shows the composition of the mixture before and after using it to oil feathers. Within the error of the analytical method, the composition is unchanged.
Direct oiling with a modified blend not only improves precision but also provides a major reduction in the cost of the hydrocarbons needed for a detergent study. The published procedure (Bryndza et al. 1991) calls for a mixture of 75 g of each of the 13 components listed in Table 5 for a total of 975 g. In May 2022, the cost for this mixture was $3500 from standard chemical suppliers, about $3.59 per g. This cost does not include the dichloromethane and the internal standard, 1-octadecene. By removing the volatile and expensive chemicals to simplify the mixture and improve reproducibility, the cost of 975 g of the mixture shown in Table 6 was reduced to $848, or about $0.90 per g (May 2022 pricing). This represents a cost savings of 75%, assuming the solution is not reused. But as has been shown earlier, it is possible to reuse the solvent blend, leading to even greater savings.
Conclusions
Revision of the previous method for objective testing of detergents for cleaning oil from feathers has resulted in a reliable procedure that greatly reduces material costs. A blind study of detergents has indicated that Palmolive Ultra®, Fairy Liquid/Dawn® Ultra Pure Essentials and Bright GreenTM (no longer available under that name) were the most effective cleaning agents for synthetically oiled snow goose feathers. This work was completed using 1% detergent solutions for the washing step. In future work, lower concentrations may be appropriate, particularly to differentiate between the most efficacious products.
An additional benefit to the revised procedure is the reduction in the quantity of dichloromethane needed for the analysis. Dichloromethane is a known animal and predicted human carcinogen (Pubchem, not dated), and care must be taken to avoid exposure by inhalation or skin contact. Whilst the amount of dichloromethane was reduced significantly by directly oiling the feathers with the hydrocarbon blend, the complete elimination of dichloromethane is an important next step, as discussed below.
Recommendations for future work
As new detergent formulations are constantly being introduced, it is recommended that a study such as this be carried out at intervals of several years. Given improvements in detergent efficacy, better differentiation between candidates may be obtained with detergent solution concentrations below 1%.
The US Environmental Protection Agency (EPA 2023) has recently proposed banning the use of dichloromethane in all consumers and most industrial/commercial use. Consequently, a new solvent for extracting residual oil from the cleaned feathers should be identified. Greener alternatives to dichloromethane include 2-methyltetrahydrofuran, ethyl acetate, toluene and methyl-tert-butyl ether (USC, not dated). Considerations for the choice of solvent include its partition coefficient with water, ability to extract oil and boiling point (so that the solvent does not interfere with GC analysis of the hydrocarbon mixture).
When presenting this work at the 2023 NWRA conference in Wilmington, DE, the authors were informed by wildlife rehabilitators that certain detergents seemed to be easier to rinse than others during cleaning of oiled birds. This study did not examine the ease of rinsing detergents from the cleaned feathers. Since the cleaning process can stress affected wildlife, any extra time for rinsing could be detrimental. Given two detergents with similar efficacy, one that rinses off more quickly would be preferred. This would be a useful consideration for future work.
Acknowledgements
The authors wish to thank the following Widener University faculty and staff for assistance with this work: Mr Martin Schultz, Dr Rob Mishur, Dr Shirley Fischer-Drowos and Dr Ismail Kul. Additional work supporting this project was carried out by Widener University students, Max Weber, James Rodgers, Justin Saporito and David McNeill. Financial support was provided by the Oiled Wildlife Care Network, Tri-State Bird Rescue & Research (TSBRR), Widener University Faculty Development and Provost’s Grants and the Widener University Summer Undergraduate Research and Creative Activities Program. DuPont generously provided several GC columns. GC supplies were purchased with a grant from Hamilton Syringe. Snow goose feathers and detergents were provided by TSBRR. Monroe Energy kindly provided authentic crude oil samples. Additionally, the authors thank Lisa Smith (TSBRR), Dr Erica Miller (TSBRR), Dr Henry Bryndza (DuPont, retired), Mr David Goshorn (Exxon, retired) and Mr Ron Bingeman (Croda, retired) for sharing their expertise. The authors also wish to thank the reviewers for helpful comments.
References
| Audubon. 2014. Accessed on the internet at https://www.audubon.org/news/more-one-million-birds-died-during-deepwater-horizon-disaster on 19 September 2024 |
| Berkner A.B., Smith D.C. & Williams A.S. 1977. Cleaning agents for oiled wildlife. In Oil spill conference. Pp. 411–415. Accessed on the internet at https://birdrescue.info/wp-content/uploads/2018/11/1977-Cleaning-Agents.pdf on 19 September 2024. |
| Bryndza H.E., Foster J.P., McCartney J.H., Lober J.C. & Lundberg B. 1995. Methodology for determining surfactant efficacy in removal of petrochemicals from feathers. In C. Rineer-Garber (ed.): Wildlife and oil spills: response, research, and contingency planning. Pp. 69–86. Newark, DE: Tri-State Bird Rescue & Research, Inc. |
| Bryndza H.E., Foster J.P., McCartney J.H., Lundberg B. & Lober J.C. 1991. Surfactant efficacy in removal of petrochemicals from feathers. In L. Frink, K. Ball-Weir & C. Smith (eds.): The effects of oil on wildlife: research, rehabilitation and general concerns. Pp. 78–94. Suisun, CA: International Wildlife Rehabilitation Council. |
| Center for Biological Diversity. undated. Accessed on the internet at https://www.biologicaldiversity.org/programs/public_lands/energy/dirty_energy_development/oil_and_gas/gulf_oil_spill/a_deadly_toll.html on 19 September 2024. |
| EPA. 2023. Accessed on the internet at https://www.epa.gov/newsreleases/epa-proposes-ban-all-consumer-most-industrial-and-commercial-uses-methylene-chloride on 19 September 2024. |
| King, M.D., Elliott J.E. & Williams T.D. 2020. Effects of petroleum exposure on birds: a review. Science of the Total Environment 755(Pt 1), 142834. doi: 10.1016/j.scitotenv.2020.142834. |
| Miller E., Keller J. & Bryndza H. 2006. An evaluation and comparison of some current products for the removal of petrochemicals from feathers. In K. Evans & R. Dunne (eds.): The effects of oil on wildlife: proceedings of the eighth international conference. Pp. 85–99. Newark, DE: Tri-State Bird Rescue & Research, Inc. |
| Miller E.A., Bryndza H., Milionis C., Meenan K. & Simmons M. 2003. An evaluation of the efficacy of eighty-six products in the removal of petrochemicals from feathers. In The effects of oil on wildlife: Proceedings of the sixth international conference. Myrtle Beach, SC. |
| Miller E.A., Bryndza H., Milionis C., Meenan K. & Simmons M. 2003. An evaluation of the efficacy of eightysix products in the removal of petrochemicals from feathers. In H. Stout (ed.): Proceedings from the 2000 Effects of Oil on Wildlife Conference, Pp. 52–66. Newark, DE: Tri-State Bird Rescue & Research, Inc. |
| Miller E.A. & Ricko A. 2022. Evaluating the efficacy of 16 surfactants for removing petrochemicals from feathers. Wildlife Rehabilitation Bulletin 39(1), 9–22. |
| Pubchem (NIH National Library of Medicine). not dated. Accessed on the internet at https://pubchem.ncbi.nlm.nih.gov/compound/Methylene-Chloride#section=Toxicity on 19 September 2024. |
| USC Environmental Health and Safety. not dated. Accessed on the internet at https://ehs.usc.edu/research/lab/sustainability-in-research-labs-at-usc/solvents-for-a-safer-sustainable-lab/ on 19 September 2024. |
| U.S. Energy Information Agency. 2023. Accessed on the internet at https://www.eia.gov/energyexplained/oil-and-petroleum-products/use-of-oil.php on 19 September 2024. |